Leadership, Infrastructure, and Investability: Neurotech's Rising Tide
Representations 02: Q1 BCI Funding Map, Roadmap, Reflections
Representations is a new limited series exploring neurotech’s evolving business frontier. This is produced in partnership with PL Neuro, a project by Protocol Labs. PL Neuro accelerates neurotechnology and neuro AI through field building, strategic investment, and scientific research. Their worldview is deeply technical at every level, yet wholly focused on enabling futures of human flourishing and empowerment. Over the coming months we’ll explore key topics, trends, and themes, featuring experts from PL Neuro’s ecosystem and market insights curated from Neurotech Futures’ platform.
Welcome back to Representations. Last time we charted BCI in the AI Era, wherein rapid advances to data flywheels and business models are unlocking upmarket applications and platform opportunities for neural interface startups.
April brought us new companies, partnerships, fundraises, authorizations, leadership moves and more across BCI. We’re sharing a new market memo that tracks and tallies commercial activity across the sector, year to date.
This edition also highlights topics in infrastructure and leadership. These critical forces are shaping the generational business opportunity to usher in a safe and sustainable cybernetic era in the coming decades.
Clinical ARR: Smoothing The Path to $100B
Protocol Labs published the first of their Brain–Computer Interface Roadmap Series this month. It’s for investors, policy makers, and strategists interested in an ambitious but clear-eyed outlook for how today’s BCI leaders could - and should - achieve collective market success for restorative neurotechnologies.
First, traditional FDA pathways to regulatory approval are increasingly misaligned with “living devices” that use adaptive AI algorithms designed to improve post-implantation.
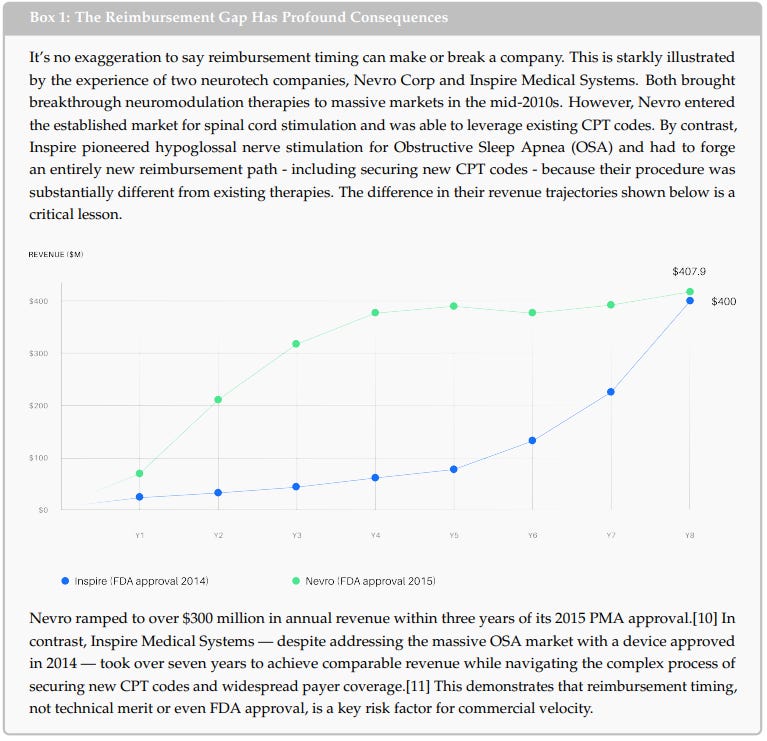
Second, the regulation-reimbursement disconnect remains a quantified structural risk that renders novel treatments inaccessible to most patients, and encourages iteration over innovation.
Third, clinical adoption — established neurotech therapies like DBS and VNS achieve only 3–9% penetration of their addressable patient populations, due to physician workflow barriers and legacy risks of traditional surgical methods and devices.
More than simply navigating the Valley of Death, the authors call for a multifaceted and concerted infrastructure building plan, with the goal of building a sustainable business for restorative BCI recipients and their supporting clinical and product teams on the device side. We’ve seen the alternative and it isn’t pretty.
The 5-year action plan has three pillars: (1) compress the FDA-CMS reimbursement gap to under 12 months by leveraging the TCET pathway and pursuing Congressional action; (2) establish shared CPT code frameworks across device classes, rather than leaving each company to negotiate codes independently; (3) create a mechanism to provide patient support if a manufacturer fails, a precondition for patient trust that no single company can credibly provide on its own.
Since this paper’s publication, the CMS & FDA’s new RAPID program and other agency developments suggest the proposed strategy could prove effective. I found the policy and industry strategies it sets forth provocative, like embedding “Neurotech Fellows” at CMS and funding an “Orphan Implant Backstop.” For aspiring (or lapsed) neurotech policy wonks, print this out and bust out the highlighter.
For investors looking for emerging markets: With systemic changes afoot at the FDA, CMS, and beyond, it is a good time to pay closer attention: BCI’s value potential is no longer speculative, aiming far beyond today’s $8.8 billion neurotech market.
Next-gen, AI-adaptive systems could drive $100B+ in annual revenue within a decade, following an adoption curve similar to how cochlear implants and DBS went from experimental to established standards of care.
Finally, the authors and advisors to this paper comprise a who’s who of innovation architects at ARIA, Astera Institute, Renaissance Philanthropy, leading venture funds, medical centers, and startups. So it’s worth skipping your next slopscroll to spin through this instead.
BCI Market Memo: Capital & Commercial Activity In 2026
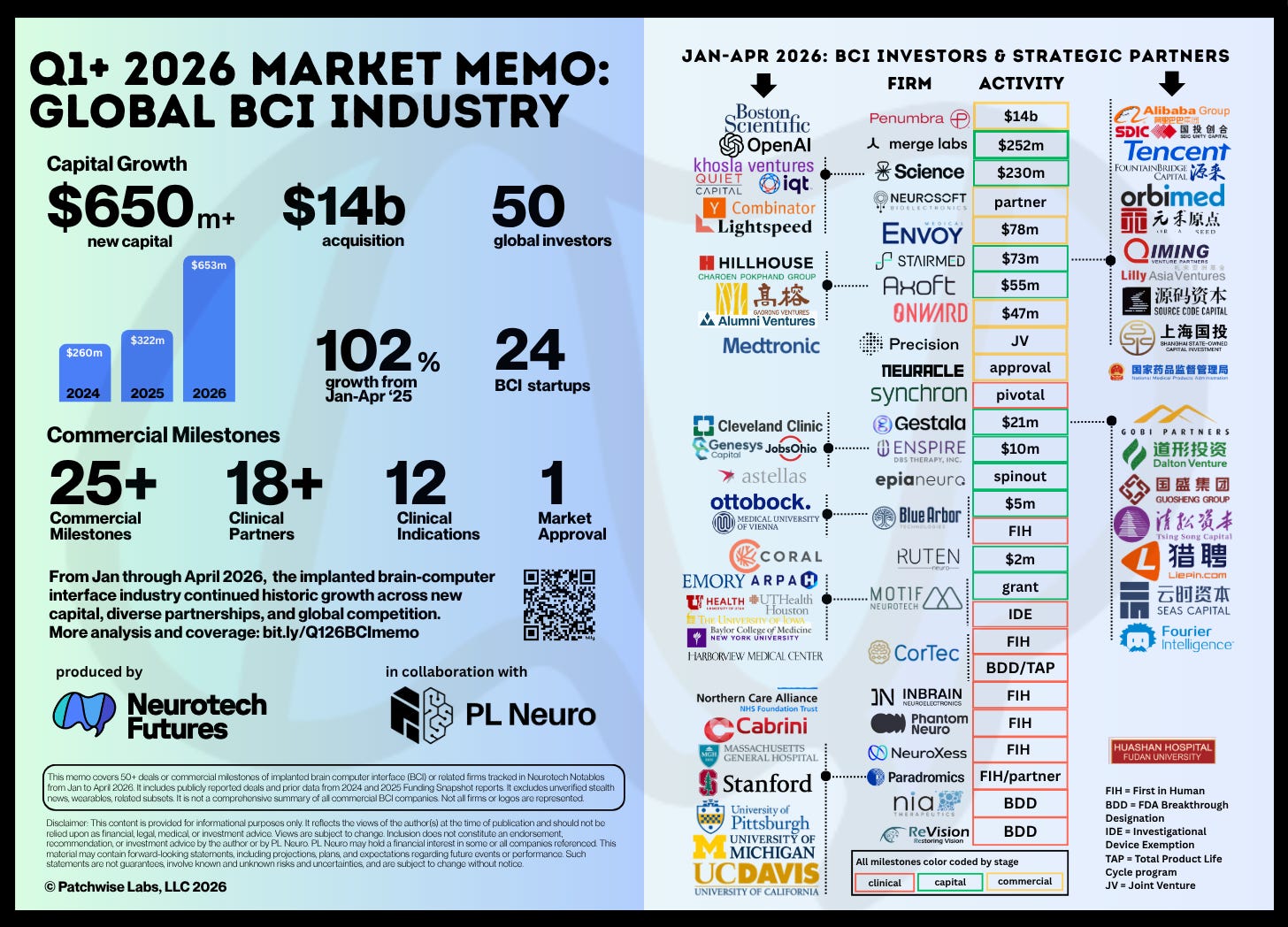
From January through April of 2026 we compiled commercial announcements across implanted BCI into this market memo. It offers a non-exhaustive, representative point-in-time analysis of the major drivers behind the field’s rise: funding growth, commercial ecosystem development, regulatory advances, and widening competition.
$653m in new capital, led by raises from Merge Labs and Science Corp, defined a historic surge compared to this same period in previous years. Yes, big rounds are still in, but to benchmark against last year: there are two months left to go in H12026 and year-to-date funding is already at 45% of last year’s $1.46b. At this pace, 2026 BCI funding will hit $2b.
Boston Scientific’s $14b acquisition of Penumbra, while not a “BCI” play, is a bet on greater neural access by a DBS company making regular neurotech acquisitions. This visual omits Mint Neuro’s May 1st deal with Motif, which adds evidence of a burgeoning commercial ecosystem following Precision-Medtronic and Science-Neurosoft deals inked in Q1.
Over 18 clinical partners launched collaborations with BCI startups for various human-stage research efforts, led by Paradromics, Motif, Cortec, and others. This year will see a slew of first-in-human implants by companies building applications across stroke, vision, limb, prosthetics, TBI, psychiatry, and speech. Synchron’s pivotal trial will also kick off this year following their $200m series D. Meanwhile, Neuracle’s commercial clearance from China’s NMPA represents a global milestone on several levels.
Chinese BCI investment crossed $100m in Stairmed, Gestala, and Axoft. The trend to monitor is the breadth of investors backing China’s BCI revolution, from regional and globally oriented venture capital firms, to family offices, state-run syndicates, and multinational corporate interests across robotics, pharma, e-commerce, manufacturing, and AI. There is much more happening in China. While beyond the scope of this memo, BCI geopolitics are of growing importance and will continue to inform global strategy across the field.
There are the usual caveats on company inclusion, categorization, and representation in this memo. Please note that any mistakes, oversights or editorial decisions were mine alone. If you have feedback, methodology questions, ideas, or just want to know more, hit reply or send me a note.
NeuroMaxxing: Juan Benet and Max Hodak discuss the next decade of the human brain
If you, like me, are intrigued about what’s going on inside the mind of Science Corp’s founder and CEO Max Hodak (one of Time’s 100 most influential people) PL Neuro’s inaugural podcast Restoring Sight, Growing Neurons on Silicon, and Expanding Human Intelligence, is just for you.
Juan adeptly weaves history and culture into questions about neurobiology and deeptech, beginning with a history of pre-clinical BCI work from the 1960’s to the development of the Utah Array at the turn of the century, through the last decade of advances in miniaturization, materials, and modernization, powered by what Hodak calls the “Smartphone Dividend.”
There is too much to summarize from the two-hour episode: Blind 81-year-olds reading newspapers and crosswords in the PRIMA trial; navigating biological constraints in neural engineering while adopting systemic and operational innovations; Max’s origin story and purview on hiring, culture, leadership; the current AI moment, and more.
The heart of this episode renews BCI’s grand vision, speculates with Juan on known and unknown sensory and cognitive enhancements, and offers a deep dive into biohybrid arrays, from neuronal biochemistry, de novo optogenetic proteins, risks, timelines, applications, and more. Like Science Corp’s secret master plan:
“Our plan, like the secret master plan here is: Bring a breakthrough retinal prosthesis to market. Use the revenue from that to fund the development of the biohybrid core technology. Use this to prove out some really big prizes of emerging neurotech, and then eventually of course translate this to market. We have this vision of how I think the world is likely to look in the early 2030s. I think 2030 will probably still look a lot like today. I mean this is also AI and other things happening, but I think 2035 is likely to look quite a bit different than we might be imagining right now.” - Max Hodak, founder and CEO, Science Corp
Make sure to subscribe to catch the next episode when it drops!
Visit PL Neuro’s growing hub of content, events, and more: https://plneuro.xyz
Disclaimer: This content is provided for informational purposes only. It reflects the views of the author(s) at the time of publication and should not be relied upon as financial, legal, medical, or investment advice. Views are subject to change. Featured projects are highlighted for informational purposes only. Inclusion does not constitute an endorsement, recommendation, or investment advice by the author or by PL Neuro. PL Neuro may hold a financial interest in some or all companies referenced. This material may contain forward-looking statements, including projections, plans, and expectations regarding future events or performance. Such statements are not guarantees, involve known and unknown risks and uncertainties, and are subject to change without notice.



